|
Bovine
Infertility
and
"Repeat
Breeder"

- A repeat breeder is defined as:
- a cow that has
calved before,
- is less that 10 years old,
- has normal heat cycles,
-
has no palpable abnormalities,
- has been bred 3 or more times and is
not pregnant.
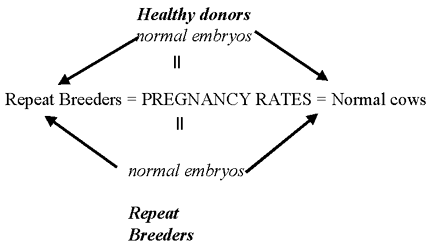
- This is a term that is apparently, and for good
reason, dying in the theriogenology literature. Researchers have
long sought the cause for repeat breeders, but have been stymied.
Why? Most likely is the methodology. Researchers gathered cows
defined as 'repeat breeders' and then looked for a specific cause.
They usually found no single cause that explained all the problems.
Not a big surprise when you consider all the possibilities in all
the cows. It is difficult to study because in 'repeat breeders',
many are cows are probably normal. Few workers have looked at
specific organisms to 'create' the syndrome. Also if you look at
fertility expectations in normal animals you see that 9% of normal
cows would be repeat breeders. We normally assume a problem exists
when the incidence is 10-15 %.
Assume 60 % conception rate
1.65 services/conception
Normal Incidence of "repeat
breeders", assuming a 60 % conception rate.
| Breeding Number |
Number Bred |
% Pregnant |
Number Pregnant |
| 1 |
100 |
60 |
60 |
| 2 |
40 |
60 |
24 |
| 3 |
16 |
60 |
10 |
| 4 |
6 |
60 |
3 |
| 5 |
2 |
60 |
1 |
| 6 |
1 |
60 |
1 |
| Total |
165 |
60 |
100 |
normal incidence to fit definition = 9 %
Ovulation failure
is rare to cause infertility.
Chromosomal/Genetic
- Inbreeding may contribute to repeat breeders.
- Known genetic causes of infertility :
- 1,29
Robertsonian translocation (most common in Simmental, Charolais,
also identified in approx. 40 other cattle breeds) ,
- 14,20 (Simmental)
- DUMPS (Deficiency of Uridine MonoPhosphate
Synthetase) Holstein) which is an autosomal recessive gene that
results in fetal death in first 2 months gestation.
- A reported XXX karyotype is associated with
infertility in Holsteins.
- ET embryos have very few chromosome
abnormalities.
- About 8.7% chromosome abnormalities have been
found in aborted fetuses and abnormal live births.
- In humans a high percent of miscarriage have
chromosomal abnormalities. This is probably because aged gametes
result from copulations that are not just at ovulation.
- Aged oocytes (i.e. 2 wave ovulations
have older oocytes than 3 wave, and 'pushing' ovulation of
small follicles)
- May result in compromised luteal
function also.
Molecular
Cytogenetics Laboratory
Room 318 B
Bldg 1197
Department of Veterinary Integrative Biosciences
Texas A&M University
College Station, TX 77843-4458
Telephone: 979-458-0520
Fax: 979-845-9972
Please call in advance. Laboratory led by Dr. Bhanu
Chowdhary
Fertilization failure
- In normal heifers you have 100 % fertilization
one day after breeding. This drops to 85 % in cows, and to 60-70 %
in repeat breeders. Therefore repeat breeders seem to have more of a
fertilization failure. If embryos are fertilized and transferred,
you have normal pregnancy rates.
Abnormal embryos
- In one study more abnormal embryos were seen in
repeat breeders, but if the embryo is normal the result is a normal
pregnancy rate.
Hormonal
- A decreased GnRH or luteinization is the basis
for GnRH treatment of repeat breeders. It has been shown that
administration of GnRH after the 3rd service helps fertility rates.
All the other studies are pretty inconclusive.
Release of PGF from inflammatory
conditions such as mastitis can cause
luteolysis and pregnancy loss
Inability to prevent PGF release (shown
experimentally in a repeat breeder in response to oxytocin
administration) causes return to estrus.
Environmental effects  403 403

- Summer
heat stress (high temperature and humidity)
- In heat stressed animals progesterone,
corticosteroids are higher and estrogen is lower that normal. Since
cows have a harder time dissipating heat that heifers, high
temperatures affect mature cows more than heifers. The smaller
nonlactating cows produce less heat and dissipate heat better,
therefore their internal temperature is lower.
- A Florida summer ET study compared AI in cows
to cow embryos placed into ET recipient heifers. The net result is
that at 45-60 days the pregnancy rate in the cows was 13.5 %
compared to 29.2 % in the ET heifers. This shows that it more of a
uterine problem than ova problem.
- Fertilization rates are normal in heat stressed
cows, but day 1 to 2 embryos are affected most by the heat. As blood
flow to uterus decreases to shunt it to the rest of the body for
cooling, the uterine temperature rises, nutrients decrease, and
waste products increase. This leads to more abnormal embryos (poorer
quality) being collected from heat stressed cows at 7 days post AI.
Treatments
- Treatments for heat stress include air
conditioning, but cost and practicality are major factors. You may
only need to cool the breeding herd (which would include peak
lactation cows), and bred cows. Work has shown that you need to air
condition 14 days before and 14 days after breeding to be effective
in controlling heat stress.
- Evaporative cooling works in low humidity
areas only.
- Wet the cows and
then blow them off to help cool them. You
must soak cows or an insulating blanket of water droplets forms.
This works best for low humidity areas.
- Shade helps, but there needs to be enough shade
to prevent the cows from crowding under the shade and becoming
hotter.
- Decrease the fiber intake. The cow's appetite
decreases so they need more concentrated energy.
- Increase the potassium in the diet to
compensate for sweat loss.
- Seasonal calving can avoid breeding in hot
weather. It seems unpopular, but many people are forced into it
anyway. Milk pricing may be problem as base pricing is adopted. In
this, a price is set on 'base' production over certain time (Aug -
Nov) and the producer receives a lower price for milk produced in
excess of "base". This is basically designed to punish
people for producing an overabundance of milk during high production
times and not enough during high demand times.

Click to enlarge
Infectious agents
 364-371 364-371
BVD
(See "Abortion" also)
- See -
R.H. BonDurant.
Theriogenology Volume 68, Issue
3, August 2007, Pages 461-473
Proceedings of the Annual
Conference of the Society for
Theriogenology, Proceedings of
the Annual Conference of the
Society for Theriogenology
- The association is unclear. But much research
has implicated seronegative cows as being prone to infertility when
exposed to the virus.
- Infection at breeding may lead to lower
fertility.
- Bovine
virus diarrhea (BVD): effects on
first trimester pregnancy
- Bovine
virus diarrhea virus can
establish itself permanently in
the host
-
“Persistently infected” (PI)
animal is the reservoir of
infection for the herd
-
Result of in utero
exposure to a BVD virus
at less than 125 days of
gestation (before the
fetus has developed a
competent immune system)
- Two major
biotypes of BVDV (based on in
vitro cell cultures)
- Non-cytopathic
(NCPB)
-
Cytopathic biotypes (CPB)
- Both
can damage conceptus
between Days 0 and 125,
- NCPB
are represented amongst PI
animals
-
Born viremic
-
Infected before they are
immunocompetent
-
No antibodies to the
NCPB strain of virus
they are carrying
- Two
biotype categories
- BVDV
I
- BVDV
II genotypes
-
IIa
-
IIb
-
Less prevalent but
associated with high
morbidity/high mortality
outbreaks
- Both
genotypes can be of either
the CPB or NCPB biotypes.
- PI
animals susceptible to mucosal
disease (MD)
- Acute and
lethal gastrointestinal and
respiratory inflammatory disease
-
“Triggered by ” conversion of
non-cytopathic biotype to
cytopathic biotype by RNA
recombination of NCPB viruses
- Sends a
PI animal into MD
- Timing of
infection
- Possible to
infect the gametes’ environment
in either the bull or the cow
- Ovary
-
Interstitial
- Luteal
- Granulosa
and thecal
-
Follicular fluid
- Oocytes (oophoritis)
- Follicles
- Endocrine
capability of the follicle had
reduced estradiol output by the
follicle
- Reduced
progesterone production by the
resulting CL
- Oocytes
-
Primordial follicles
- Primary
follicles
- Secondary
follicles
- Infected
embryos are destroyed
-
Uninfected embryos become
uninfected fetuses and survive
to term without ill effects
- Several
reports exist of bulls with BVDV-positive
semen
- most caeses,
the bull is a PI animal infected
with NCPB BVD
- CP or NCP
biotypes of virus induced the
loss of the conceptus when
inoculated into cows at
approximately 30 days
- Animals
viremic with NCPB at the time of
AI - conception rates that were
similar to controls, but
significantly reduced embryonic
survival to Day 77
-
Within- and between-biotype
and genotype strain
differences
- Day 42 to
about Day 125
- Placentomes
are well established,
- Fetal
infection is likely to be the
result of placentome infection
and associated vasculitis
following a period of maternal
viremia
- Infection
can lead to abortion,
developmental defects (defects
of the derivates of the
embryonic neurotrophectoderm)
- NCP BVD
only - PI calf born viremic and
without circulating antibodies
to the infecting strain.
- Diagnosis
- Directed at
detecting antigen.
- Virus
isolation - gold standard
- PCR
-
Immunofluorescent assays from
buffy coats
-
Immunohistochemistry (IHC) of
skin taken from an ear notch
- Advantages
of the skin-punch IHC:
- Simplicity
of sample collection
- Efficacy
in formalin-preserved tissues
- Employs a
monoclonal antibody against BVDV
that can detect a diverse array
of isolates
- Antigen
capture ELISA of homogenates of
ear notch tissue, buffy coats,
or serum - good for most.
- Many
assays sensitive enough to
identify PI animals, but few
have the sensitivity necessary
to detect transiently (acutely)
infected animals.
- Positive
results with the screening tests
should be confirmed by virus
isolation.
- Serology
- Surveillance
tool to:
- Assess
vaccine efficacy
- Client
compliance
- Confirm
exposure after observation of
clinical signs suggestive of BVD
- Sentinel
animals (seronegative calves
after their colostral titers
have waned).
- Document
acute infections in non-PI
animals
- ELISA that
can detect BVD-specific IgM
antibody will provide evidence
of recent exposure
- Pre-colostral
seropositive, virus-negative
calf born to a dam acutely
infected in late pregnancy, is
unlikely to be a threat to other
animals, but is a sign that the
virus is active in the herd.
- Control
- Europe- low
prevalence of BVD virus
infection, a test-and-slaughter
program and complete eradication
is near
- North
America, where prevalence is
much higher, a more complex
approach to control is needed
- “Closed
herd”
-
Identification and culling of PI
animals
- Main
reservoir for BVDV in a herd
is the PI animal
-
Vaccination alone almost
certain to cause
disappointment
-
Identifying PI animals
difficult
-
National prevalence is
thought to be 0.5–2.0%
-
Have to check nearly 200
animals to find the one
that needs action.
-
Pooling strategies
-
Individual
animal samples can
be pooled
-
Examined
with one of the
diagnostic assays
discussed
-
Antigen
capture of extracted
ear notch samples,
buffy coats or sera,
or PCR of buffy
coats lend
themselves to
pooling
-
Skin punch IHC assay
does not lend to
pooling
-
IHC method will
yield the necessary
information on
single test.
-
Assays that detect
whole virus or viral
nucleic acid must be
run twice to
distinguish PI from
transiently infected
- Using
serology in sentinel animals for
surveillance to eliminate the
need for vaccination
-
Immunization in North America
-
Because the virus is
maintained in a herd via PI
animals, it is unlikely that
vaccination alone will
achieve the protective state
desired
- In
combination with
identification and removal
of PI animals, vaccination
can limit the effects
- Vaccines
- 180
licensed BVD immunizing
products
-
Modified live
-
Killed vaccines
-
Vaccines containing various
combinations of Type I and
II genotypes
-
Vaccines containing only CP
biotypes
- Only
NCP biotypes
-
Combinations of CP and NCP
-
Genotype I antigens protect
against type I and to a
slightly lesser extent,
against type II
- Type
II virus only does not
protect against type I
-
Ability of the vaccine to
protect the host
-
Ability to protect the
conceptus throughout
gestation
- Fetal
protection “intercepts” BVDV
before viremia and /or
population of the
placentomes
-
Cell-mediated immunity and
humoral immunity both active
in preventing the virus from
infecting the placentome.
IBR (See
"Abortion" also)
- Infectious pustular vulvovaginitis (IPV) has
been shown to cause infertility. Vaccination can cause a transient
oophoritis and result in infertility
Ureaplasmas
- Ureaplasms cause a granular vulvovaginitis by
colonization of the vulva and vagina.
- The conception rate falls to 28 % in
the acute
phase.
- It is seen mostly in Canada and the Northern
states.
- The diagnosis is made by clinical signs and
culture.
- Treatment is by using a double sheathed straws
and post breeding infusions of oxytetracycline.
Mycoplasmas are
similar to ureaplasmas.
- M. bovis causes infertility, endometritis, and
salpingitis.
- M. bovigenitalium causes an occasional
abortion.
- Acholeplasm has the same signs as same as
ureaplasm.
- These organisms can be cultured from normal
animals though.
Chlamydia
- There is no established proof that it causes
infertility, but is has been isolated from slaughterhouse cows with
pathologic lesions.
Q fever and
Bluetongue
- They may cause
repeat breeders and anestrus, but almost all cows seropositive, so it is
hard to prove.
Leptospirosis
-
Agent
-
L. interrogans
serovar hardjo. - was considered the most
important of the antigens
-
Leptospira now
classified into “genome species” based on genetic
sequences
-
“Lepto hardjo” is
actually Leptospira interrogans, serovar
hardjo type hardoprajitno, (found primarily
in the British Isles).
-
US cattle exposed to
a different species Leptospira borgpetersenii,
serovar hardjo, type hardjobovis.
-
The two organisms
(L. interrogans serovar hardjo and
Leptospira borgpetersenii, serovar hardjo,
type hardjobovis )share significant surface
antigens used for serological typing
-
Different
genomically. The L. hardjobovis genome has
been markedly reduced, suggesting that it has lost
some of the genetic potential to adapt to new hosts
-
For simplicity,
L. hardjobovis will be used to denote L.
borgpetersenii serovar hardjo type
hardjobovis here.
-
L. hardjobovis
causes frank abortions, increased infertility (reduced
conception rates) associated with carrier cows and and
bulls.
-
Diagnosis
-
Treatment
- Carrier state exists in the
kidney, so need to clear the carrier state
- Older protocols - combinations of
penicillin and streptomycin
- Prohibitive milk and
meat withdrawal times
- Availability of
dihydrostreptomycin
- Restriction on the use of
aminoglycosides
- Newer
- A single injection of
oxytetracycline (20 mg/kg, i.m.); not for use in
lactating dairy cows.
- A single injection of
Tilmicosin (10 mg/kg, s.c.); not for use in
lactating dairy cows.
- Multiple injections of
ceftiofur (i.e., 5 mg/kg, i.m. once daily for 5
days; or 20 mg/kg, i.m. once daily for 3 days); for
withdrawal times, see Food Animal Residue Avoidance
Databank [FARAD:
http://www.farad.org/].
- east impact on milk or
meat withdrawal
- Dose above the labeled
dose so need milk withdrawal
- Two injections of amoxicillin
(15 mg/kg, i.m. twice at a 48 h interval); 96 h milk
withholding time and 25 days meat withholding time.
- Vaccine
- Monovalent vaccines provoke
cell-mediated Th1-type responses
- effective defense against
abortion or reestablishment of the carrier
state, against infection
- Multivalent vaccines do
not induce this type of response.
- Two monovalent vaccine
antigens cross react with serovar grippotyphosa
'Pathologic causes '
White heifer disease
(segmental aplasia)
- These cows may be anestrus or have enough
normal uterus that they cycle, but enough of the uterus or oviduct
is missing so they do not get pregnant.
Salpingiitis
- Salpingitis is difficult to diagnose.
- 50% of
cows with palpable lesions have patent oviducts
- 50% of cows
with non patent oviducts have no palpable lesions.
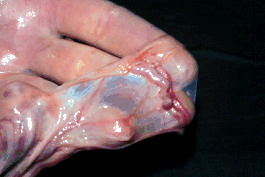
- Oviduct patency tests such as the PSP and
starch grain test are no good for diagnosis.
Metritis and cervicitis
can cause a change in the uterine environment that leads to infertility.
Post breeding infusion in these cows is not routinely
helpful.
Nutritional
- Post partum nutrition is most important for fertility
- If TDN is Low both prepartum and postpartum,
fertility suffers.
|
pre partum
|
post partum
|
conception rate % |
|
|
HI
|
HI
|
95 |
|
|
HI
|
LO |
77 |
|
|
LO
|
HI
|
95 |
|
|
LO |
LO |
20 |
|
-
Vit A had no effect on fertility, but may cause
irregular cycles.
- Vit D deficiency suppresses signs of estrus and
delays ovulation.
- Vit E deficiency may cause reproduction
problems.
- If the BUN is greater than 20 mg/100 ml cows
may have low conception rates . The high BUN is from excess dietary
protein.
|




 Bovine
Index
Bovine
Index 
